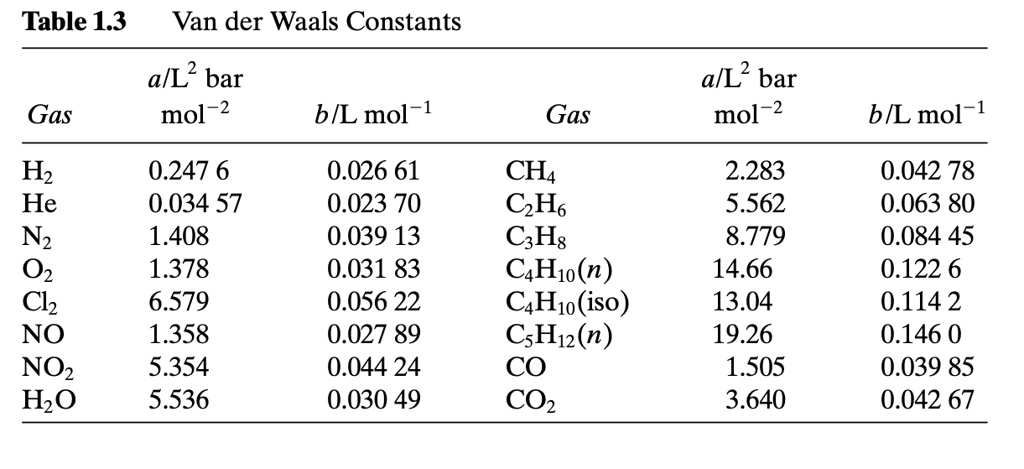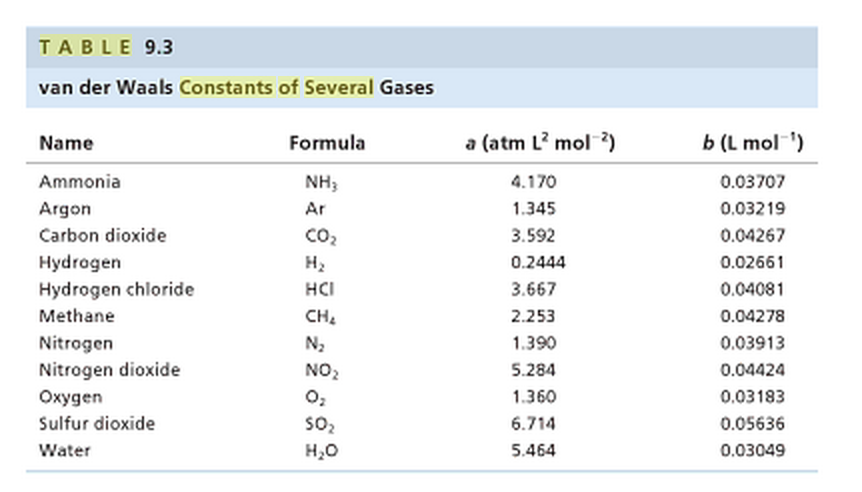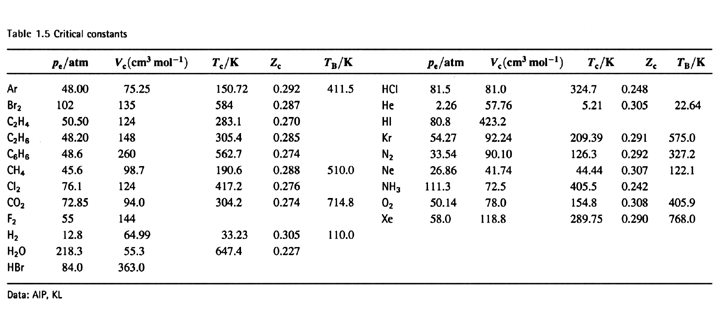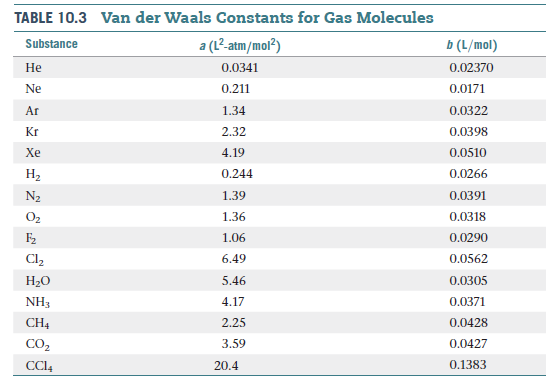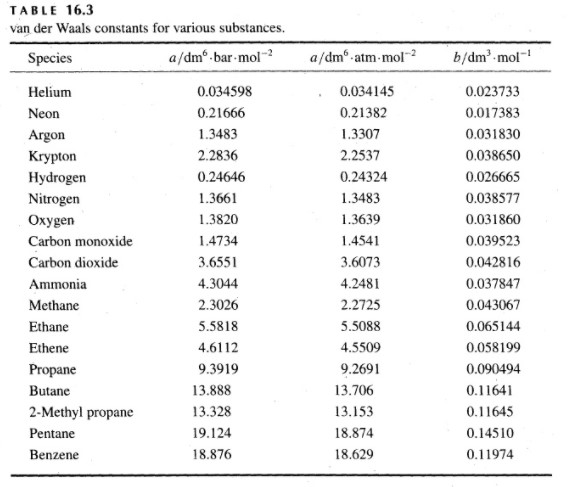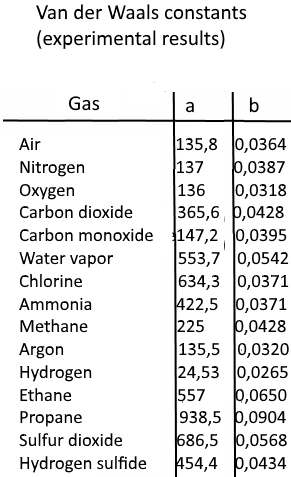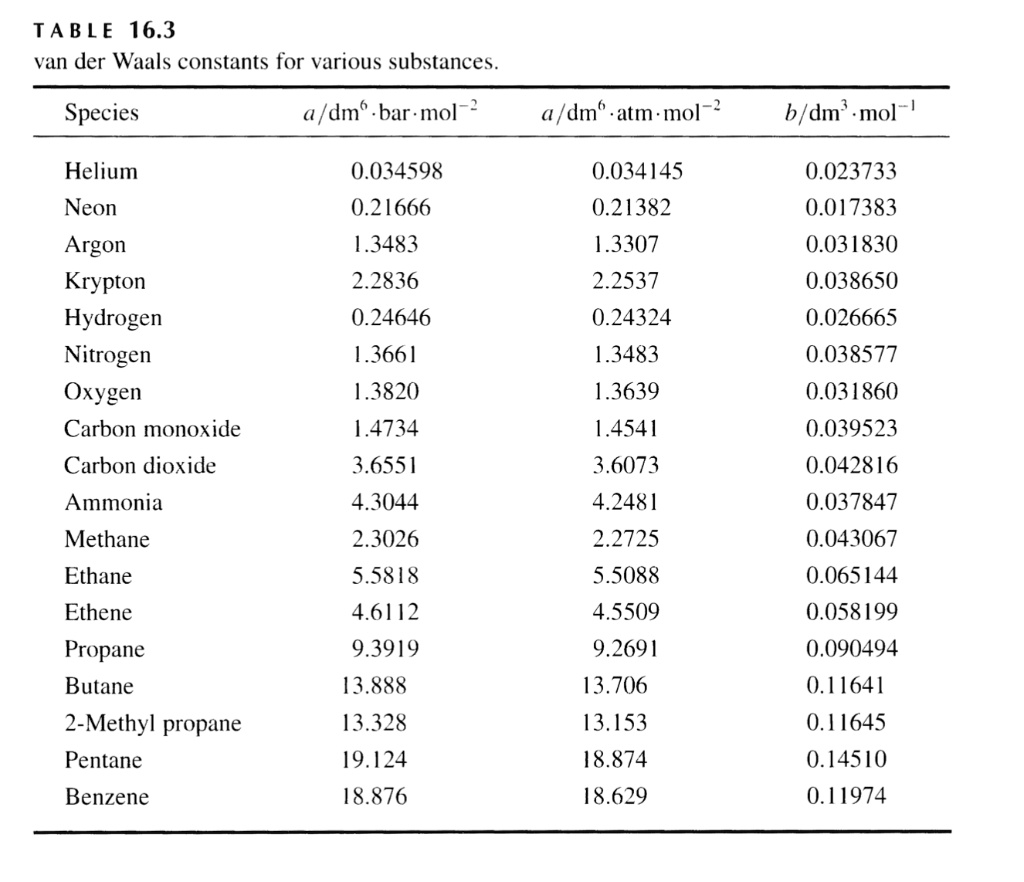
SOLVED:TA BL E 16.3 van der Waals constants for various substances Species /dm6 . bar-mol dm" . atm- mol b/dm'. mol Helium Neon Argon Krypton Hydrogen Nitrogen Oxygen Carbon monoxide Carbon dioxide

physical chemistry - Why does small value of van der Waals gas constant "b" ensure easier liquefication? - Chemistry Stack Exchange

For real gas van der Waals equation is written as: ( p + an^2V^2 ) ( V - nb ) = nRT Where a and b are van der Waals constants.Two sets

The ratio of van der Waals' constants a and b, has the dimension of (a) atm ? (C) L mol-1 (b) L atm mol-1 (d) atm L mol-2

At T=300K, 1.00mol of CO2 occupies a volume of 1.50L. Calculate the pressures given by the ideal gas equation and the van der Waals equation. (The van der Waals constants a and

OneClass: Based on the van der Waals equation of state and given the van der Waals constants below, w...
Which of the following expressions represent the value and unit of van der Waals' constant a? - Sarthaks eConnect | Largest Online Education Community

11111 Umu) 32 min 46. The ratio of van der Waals' constants a and b, has the dimension of lá atm L- ((b) L atm mol-' (c) L mol-1 (d) atm L

The van der Waals constant for gases A, B, and C are as follows Answer the following: Which gas has the